Example Of A Weak Electrolyte
Electrolytes are substances that become ions when they get dissolved in water.
INTRODUCTION:
Where practice we utilise electrolytes in daily life? Take a estimate. Don't nosotros utilize salt in our food? That's an electrolyte. Those special sports drinks offered to athletes are as well electrolytes. The detergent nosotros use for washing our clothes is once again an electrolyte. These are a few instances wherein electrolytes play an important role in our day-to-day lives.
 Source
Source
Just THE QUESTION ARISES: WHAT IS AN ELECTROLYTE?
Electrolytes are substances that become ions (an atom or molecule with a net electric accuse) when they get dissolved in water and acquire the chapters to comport electricity. A positively charged ion is called a cation, whereas a negatively charged ion is called an anion.
Based on ionization (dissociation into constituent ions) in an aqueous solution, electrolytes are of 2 types: strong and weak.
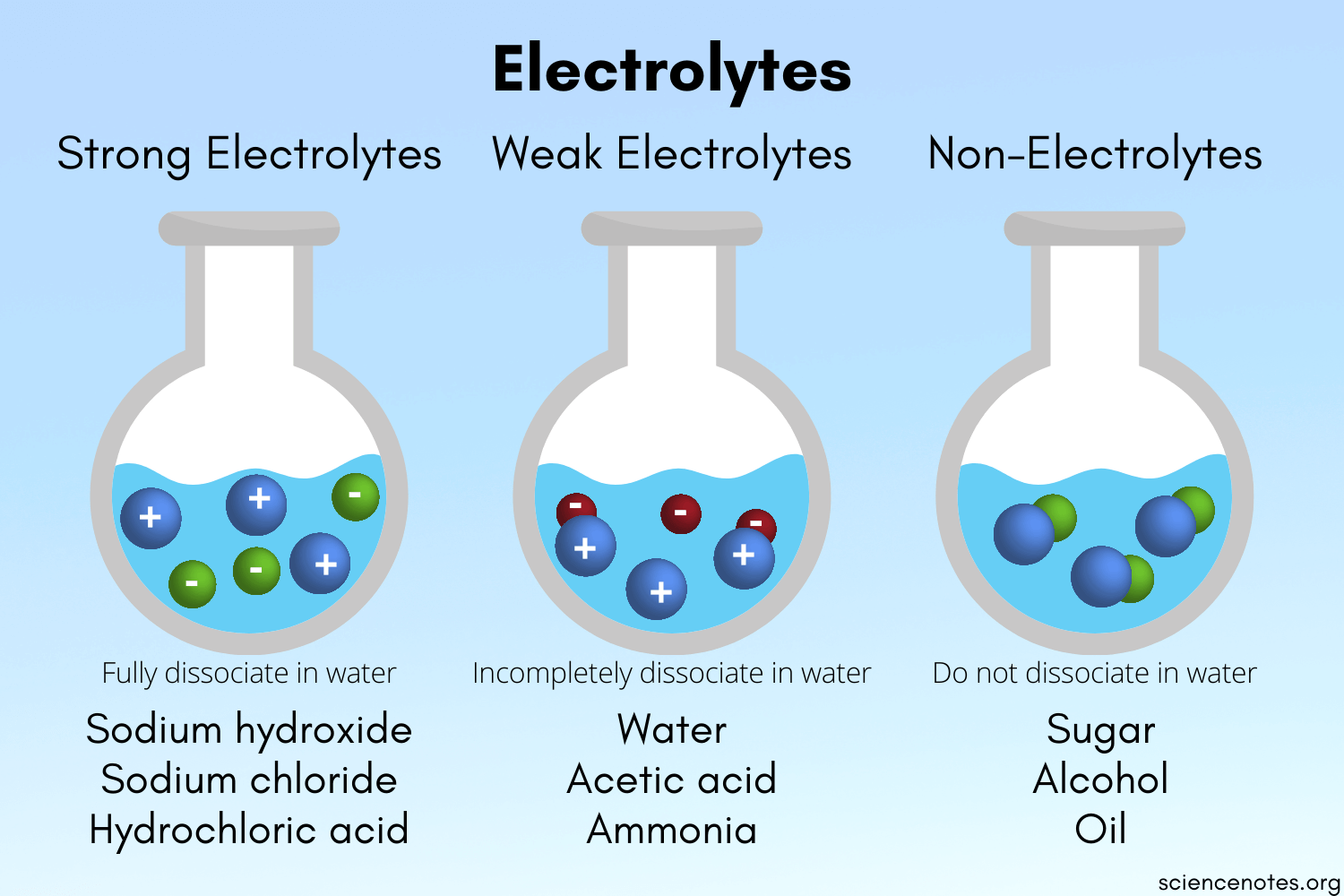 Source
Source
STRONG ELECTROLYTE:
Strong acids, bases, and salts are examples of strong electrolytes. In an aqueous solution, these compounds dissociate into ions.
EXAMPLES:
- HCl - Hydrochloric acid
- HBr - Hydrobromic acid
- How-do-you-do - Hydroiodic acid
- NaOH - Sodium hydroxide
- NaCl - Sodium chloride
WEAK ELECTROLYTE:
In water, weak electrolytes but partially disintegrate into ions. Weak acids, bases, and a range of other substances are examples of weak electrolytes. The majority of nitrogen-containing substances are weak electrolytes.
EXAMPLES:
- HF - Hydrofluoric acrid
- CH₃CO₂H - Acerb acid
- NH₃ - Ammonia
- H₂O - H2o
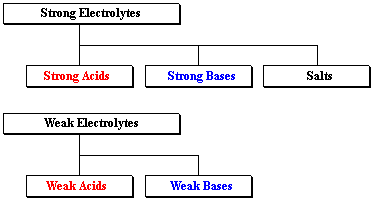 Source
Source
Conclusion:
- An electrolyte is a substance that dissolves in water and acquires the capacity to conduct electricity.
- There are two types of electrolytes- strong and weak electrolytes.
- A stiff electrolyte is an electrolyte that dissolves virtually completely in h2o. An example of a strong electrolyte is Hydrogen Chloride (HCl).
- A weak electrolyte is an electrolyte that doesn't dissolve completely in h2o.
FAQs:
1. Is HCl acid a strong electrolyte?
Aye, hydrogen chloride is a strong electrolyte, and it dissolves in h2o completely to form Hydrogen ions and chlorine ions.
two. Why is HCl an electrolyte?
HCl is an electrolyte because it completely dissolves to form hydrogen ions and chloride ions when mixed with water. These together conduct electricity. Hence, information technology's an electrolyte.
3. When does HCl act equally a non-electrolyte?
In its pure form, HCl gas is a non-electrolyte. Electrolytes are formed one time it's dissolved in water. Since hydrogen chloride gas is not dissolved in water, it's a non-electrolyte.
We hope you enjoyed studying this lesson and learned something cool about Strong and Weak Electrolytes! Join our Discord community to get whatsoever questions you may have answered and to engage with other students but like you! Don't forget to download our App to experience our fun, VR classrooms - we hope, information technology makes studying much more fun! 😎
SOURCES:
- Strong and Weak Electrolytes. https://www.ck12.org/c/chemical science/potent-and-weak-electrolytes/lesson/Stiff-and-Weak-Electrolytes-CHEM/ accessed 17 Feb 2022
- Chemical science Examples: Potent and Weak Electrolytes. https://www.thoughtco.com/strong-and-weak-electrolytes-609437 accessed 17 Feb 2022
Example Of A Weak Electrolyte,
Source: https://www.inspiritvr.com/general-chemistry/water/strong-and-weak-electrolytes-study-guide
Posted by: jewellcracenduch.blogspot.com


0 Response to "Example Of A Weak Electrolyte"
Post a Comment